Graduate School of Science and Engineering Science of Environment and Mathematical Modeling
- Course Outline
- Earth System Science / Environmental Magnetism Laboratory
- Geo-environmental Science Laboratory
- Wild Life Preservation Laboratory
- Advanced Materials Science and Process Systems Laboratory
- New Energy System Laboratory
- Environmental Systems Engineering Laboratory
- Regional Environment Laboratory
- Geometry Laboratory
- Functional Equations Laboratory
- Statistical Finance Laboratory
- Computational Mathematics Laboratory
- Laboratory of Mathematics for Information
- Discrete Mathematics Laboratory
- Algebra Laboratory
- Analysis Laboratory
Environmental Systems Engineering Laboratory
Smart Materials for Energy and Environmental Issues
Staff

MORIMITSU Masatsugu
[Professor]
| Acceptable course | |
|---|---|
| Master's degree course | ✓ |
| Doctoral degree course | ✓ |
Telephone : +81-774-65-7765
mmorimit@mail.doshisha.ac.jp
Office : KE-409
Database of Researchers
Research Topics
- Rechargeable Metal Hydride/Air Battery (HAB)
- Highly Sensitive and Selective Ion Sensor
- Smart Anode for Electrowinning, Electroplating, Water Electrolysis, and Waste Water Treatment
Research Contents
<1> Rechargeable Metal Hydride/Air Battery
Energy storage and supply system linked to renewable energies such as solar and wind powers is one of the most important infrastructures to reduce CO2 emission in generation and consumption of electric energy, and a secondary battery is the key technology of electric energy storage and supply. Advanced battery development beyond lithium ion secondary batteries (LIB) has been carried out by academia and industries in past decades, in which a high energy density (ED) beyond LIB is required, which means electric energy to store per unit volume or weight of a battery is high. Materials needed for high energy density are mostly chosen from more reactive ones than lithium in combination with flammable solvents for the electrolyte, although high energy density storage devices should keep themselves safe and environmentally friendly.
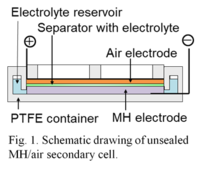

We have been developing a new class of secondary air battery with the bi-functional air electrode and focusing on hydrogen storage alloys as the negative material in combination with an alkaline electrolyte. This secondary battery is expressed as metal hydride/air battery (HAB), which is similar to the configuration of Ni-MH battery where the nickel oxide electrode is replaced with the air electrode (Fig. 1 and Fig. 2). The battery has a high theoretical energy density more than 1,000 Wh/kg or 3,000 Wh/L, although the EMF is 1.22 V. Our recent studies have demonstrated that the current efficiency and the utilization of MH are higher than 90% and the energy density is about 900 Wh/L. The battery is expected as a promising candidate for use in solar and wind power storage and power supply.
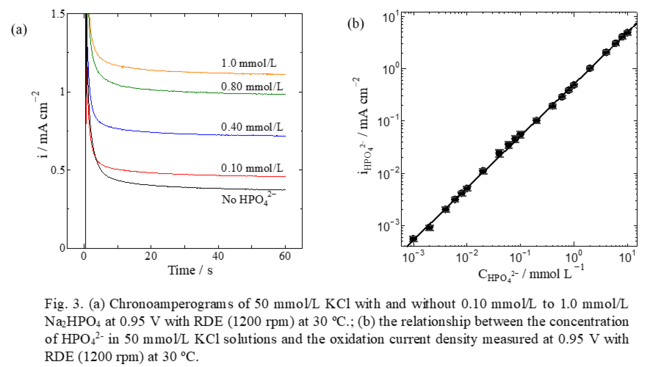
<2> Highly Sensitive and Selective Ion Sensor
Compositional analysis of blood and urine is important for illness diagnosis and health management, and electrochemical (EC) ion sensors are one of the useful devices for them. EC sensors have a potential of rapid, highly sensitive, and highly selective detection and quantification, which strongly depends on the material and structure of the detecting electrode. Our laboratory has been developing smart electrodes for such ion sensors and has demonstrated to be able to quantify hydrogen phosphate and hydrogen carbonate in blood with the smart electrode comprising amorphous mixed oxides. The smart electrode has shown a linearity between the concentration and the oxidation current density of the ions over 4 digits of concentration range (Figs. 3). The smart electrode is also appropriate to ion sensing of those ions in environmental water.
<3> Smart anode for electrowinning, electroplating, water electrolysis, and waste water treatment
Electrowinning and electroplating of metals, water electrolysis, waste water treatment and other industrial electrolysis consume huge electric power, and the energy consumption depends on the voltage of the electrolytic cell, in which the electrolyte is usually an aqueous solution and the anode's reaction is oxygen or chlorine evolution. A low overpotential anode is significant for energy saving, and the smart anode developed by our group can accelerate the anode's reaction and reduce oxygen or chlorine evolution potentials. The anode consists of amorphous oxide formed on a titanium substrate, which is prepared by thermal decomposition of a precursor solution. The amorphous oxide anode can reduce oxygen evolution potential by 700 mV for copper or zinc electrowinning compared to the lead alloy anode.
Keywords
- Smart Materials
- Electrocatalyst
- Rechargeable Air Battery
- Ion Sensor
- Smart Anode
- Electrowinning
- Electroplating
- Water Electrolysis